 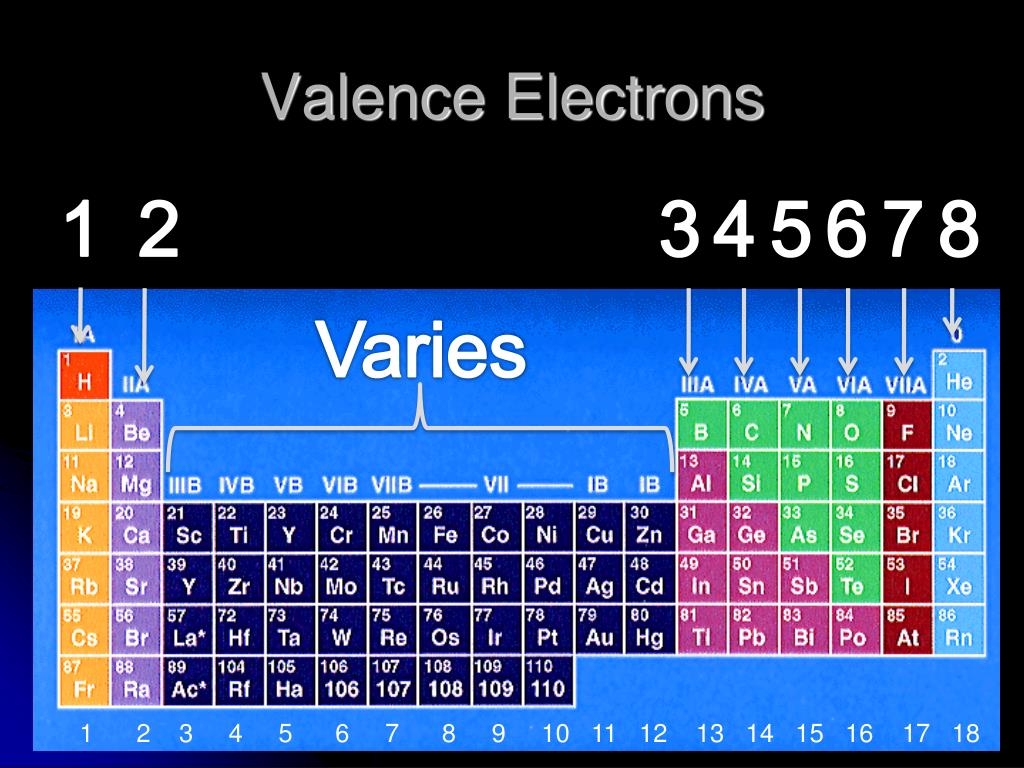
The classification of elements by atomic number allows us to understand many properties of the atom and makes it possible to predict behaviors instead of just having to memorize everything. 209, indicates the mass number of the longest-lived isotope of the element. 
(Credit: User:Cepheus/Wikimedia Commons Source: (opens in new window) License: Public Domain) Mass numbers of typical isotopes of Silicon are 28 29 30. Since 1971, SI defined the 'amount of substance' as a separate dimension of measurement. Thus, for example, the molar mass of iron is about 55.845 g/mol. Classified as a metalloid, Silicon is a solid at room temperature. In a typical sample of carbon-containing material, 98.89 of the carbon atoms also contain 6 neutrons, so each has a mass number of 12. For chemical elements without isolated molecules, such as carbon and metals, the molar mass is computed dividing by the number of moles of atoms instead. Silicon is a chemical element with symbol Si and atomic number 14. The element carbon (C) has an atomic number of 6, which means that all neutral carbon atoms contain 6 protons and 6 electrons. \): The periodic table classifies elements by atomic number. Mass Number(A) Number of Protons + Number of Neutrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
